Science is promising us steak that’s heart-healthy, eco-friendly, and still decadent. But will we eat filet mignon from a bioreactor?
Article published at www.townandcountrymag.com on May 2, 2021.

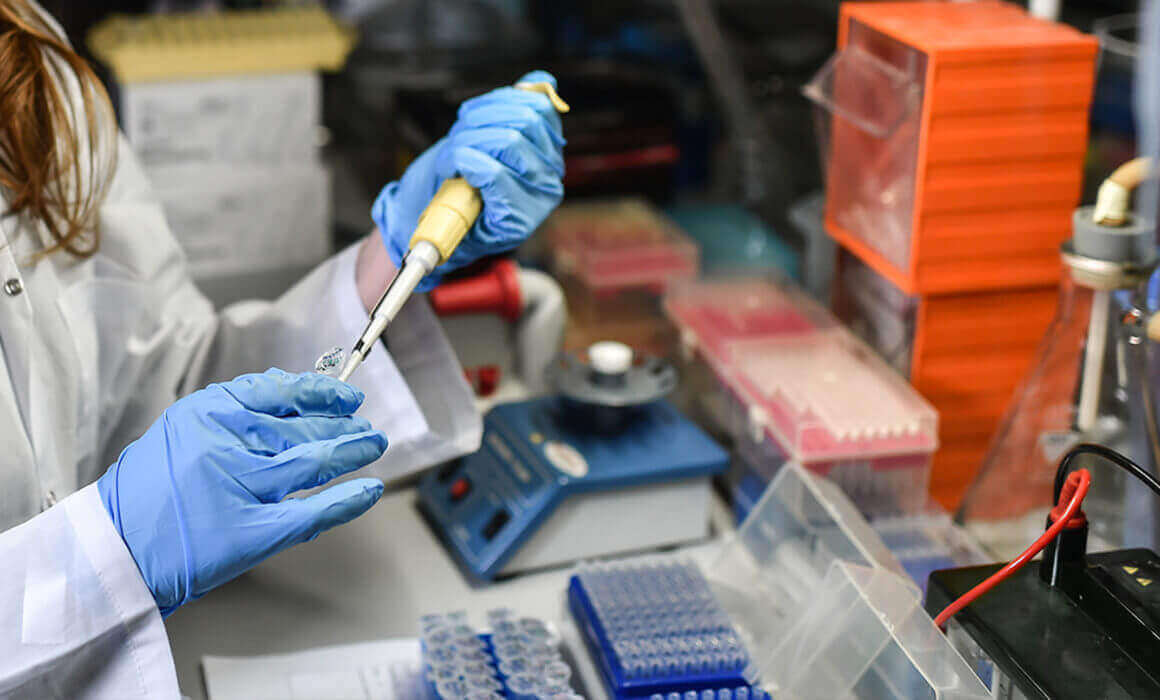
On the third floor of a modern marble and glass building half an hour from Tel Aviv and around the corner from the Weizmann Institute of Science, Israel’s leading research institute, Neta Lavon snaps on a pair of blue latex gloves and opens an incubator. She removes a plate of six circular dishes covered with clear plastic, each containing a clear solution of vitamins, minerals, amino acids, and all the other compounds that the cells she is working with need to grow. Lavon, 49, a biology PhD who earlier managed the human embryonic stem cell lab at Cedars-Sinai Research Center in Los Angeles, takes a seat on a rolling chair before a large box about nine feet tall, six feet wide, and two feet deep. The box has a plastic screen like the ones we have grown accustomed to seeing at banks and checkout lines; here, it covers a shelf holding vials, pipettes, and other paraphernalia of scientific labs the world over.
“When I want to give the cells fresh nutrients, I take out the used growth medium and replace it with fresh,” Lavon says. The box, called a biological safety cabinet, washes the shelf with filtered air so she can slip her hands through an opening in the screen and uncover the dishes without contaminating them with the tens of millions of microbes that float around us every minute of every day. She exchanges the growth medium before moving the re-covered plate to a microscope, where she can check on the cells’ progress.
A computer screen shows what’s under the lenses: dozens of gray-white cells, roughly triangular in shape, with black nuclei, in a gray solution. “Every 24 hours they duplicate themselves until they fill the whole surface,” Lavon says. “Then we harvest the cells—that’s the terminology we use—and expand them.”
Once enough cells have developed, they’re placed in a bioreactor, a vessel for carrying out a biological process similar to devices the pharmaceutical industry uses to manufacture vaccines. The one in Lavon’s lab, a chrome-plated cylinder about a foot tall with tubes sticking out of it, looks like a blender on life support. A motor on top mixes the cells so they replicate in suspension.
“In our pilot plant we are going to have vessels that are 1,000 or more times bigger than this one,” says Lavon. “They can grow into a big mass of cells that then we can make into the meat.”
Wait, what? Meat?
The New Butcher Case
Lavon is vice president of research and development at Aleph Farms, which in 2018 produced the first steak without the use—or, many say, abuse—of a living, breathing animal. That was a thin-cut steak—not exactly a hallmark of chefs or backyard grillmasters—but it was merely a stepping stone to Aleph’s announcement in February that it had developed a ribeye, considered by many the finest cut of beef.
Aleph is one among an expanding field of companies racing to bring to market what they would rather not be called “lab-grown meat” (they prefer “cultivated” or “slaughter-free”). Though the technology did not exist even just a few years ago, today at least 33 startups in 12 countries are producing a variety of meats—from dog food to foie gras, pork to duck, chicken nuggets to beef patties. Some are promising cultivated meat in stores next year.
33 companies currently grow meat in labs—from dog food to foie gras, pork to duck, chicken nuggets to hamburger patties.
Investment is also growing rapidly: At the end of 2019 companies had raised $166 million; early in 2020 Berkeley, California’s Memphis Meats took in $161 million on its own. (Aleph has raised $14 million from Big Food heavyweight Cargill, the Israeli Innovation Authority, and others.) Together they will be rushing into a space that environmentalists, the health-conscious, and promoters of wellness have cultivated carefully. Vegetarianism is growing, from 1 percent of the U.S. population to 6 percent in recent years, as is the “flexitarian” diet.
Animal welfare is one reason many choose vegetarianism or veganism. Others aren’t ready to take that step, but, as you’ve no doubt noticed if you’ve ever set foot inside a Whole Foods, they are willing to pay more for beef that is grass-fed—meaning no “finishing” on corn in concentrated animal feedlot operations, where close confinement provides a less than ideal lifestyle for the residents—or for eggs and chicken that are free-range, even though no regulatory standard exists for such terms.
 Aleph Farms co-founder Didier Toubia holds a plate with a “traditionally” grown steak in one hand and petri dish containing cow cells in the other. PICTURE ALLIANCE
Aleph Farms co-founder Didier Toubia holds a plate with a “traditionally” grown steak in one hand and petri dish containing cow cells in the other. PICTURE ALLIANCE
Restaurateurs have noticed these trends. “There is no doubt that diners are increasingly interested in nonmeat offerings,” says Dan Kluger, owner of Loring Place, in Greenwich Village, and veteran of some of the country’s most lauded kitchens. “They have concerns about the environmental…” he trails off. “But protein grown in labs? I’ve just been trying to get people to eat more vegetables.”
Bill Gates, who along with Richard Branson, Cargill, and Tyson Foods was an investor in Memphis Meats’ series B round of investment, told MIT Technology Review in February that to “avoid climate disaster,” rich countries like the U.S. “should move to 100 percent synthetic beef” (which would be good for Memphis Meats, and hence good for Bill Gates). That seems unlikely, given the more than 10,000 years of raising livestock ingrained in our civilization, but cultivated meat will find a niche because the march of science has not been good for cattle producers.
 ILLUSTRATION BY JOE DARROW
ILLUSTRATION BY JOE DARROW
The first studies on the effects of cholesterol, of which there is a copious amount in beef, were published in the 1940s; by 1964 medical journals were replete with warnings, such as the one in Angiology that year that told of “considerable indications for the supposition that the risk for the development of coronary disease is enhanced by hypercholesterolemia [high cholesterol].” The American Heart Association today recommends no more than 13 grams of saturated fat per day for people watching their cholesterol intake; a single McDonald’s Quarter Pounder with Cheese contains 12.
At the same time, researchers have been painting an increasingly bleak picture of the effects on the global climate of producing meat in the conventional (whoops, Aleph prefers “traditional”) way. Livestock for beef and milk production are responsible for about 10 percent of humanity’s greenhouse gas emissions—more than two-thirds the amount produced by the transport sector. The environmental effects of raising cattle can be both direct (methane released as part of the digestive process is the second-most abundant anthropogenic greenhouse gas, after carbon dioxide, and 25 times as effective as CO2 at trapping heat) and indirect (nitrogen fertilizer that grows corn that is fed to cattle spills into waterways and flows into oceans, where the nitrogen crowds out oxygen molecules that fish need to survive. Eating beef, apparently, impedes our ability to eat fish).
And we’re going to need a lot more of both. The world’s population is expected to reach 9.8 billion by 2050, a 30 percent increase in as many years. “I don’t think we can keep producing and eating animals in this manner much longer,” says Daan Luining, founder and chief technology officer of Meatable, a cultivated meat startup in the Netherlands. “We’re going for big impact—which is what’s needed in terms of climate change, antibiotic resistance, animal welfare.”
In early 2020 Memphis Meats, just one of more than 30 international companies developing “cultivated” raised $161 million from investors.
Luining, Lavon, and their competitors hope to produce meat much more efficiently and, in the process, reap environmental benefits. The Good Food Institute, a Washington, DC–based nonprofit that advocates for a food system that’s “better for the planet, people, and animals,” claims that cultured beef will use 95 percent less land than livestock and cut climate change emissions and nutrient pollution from beef production by three-quarters and by 94 percent, respectively.
The coronavirus pandemic has brought into stark relief one more argument for advancing technology to supply our hunger for meat. The virus that causes Covid-19 is widely believed to have evolved from one that circulated for years in another mammal. As the virus replicated, mutations developed that enabled it to infect and sicken humans. Thanks to our palates, Americans don’t generally eat bats, the animals most widely suspected of harboring SARS-CoV-2’s precursor, but two other potentially fatal viruses, the influenza strains H1N1 and H5N1, have come from poultry and livestock in recent years—suggesting that more are on the way.
And if pandemics aren’t enough to convince people, maybe antibiotic resistance is. Cattle producers discovered some time ago that giving their animals antibiotics to head off any possibility of bacterial infection also causes even healthy cattle to grow faster. Today meat producers in the U.S. are the largest purchasers of antibiotics—more than the healthcare industry. Overuse of antibiotics has accelerated the evolution of bacteria that can resist them, and now around 700,000 people all around the world die every year from what should be treatable infections.
Grill Marks
In March, Aleph’s in-house chef, Amir Ilan, gave me a video cooking demonstration with one of the company’s thin-cut steaks. Dressed in an apron and standing in his kitchen in Israel, he heated a cast iron pan on a black electric range while sautéing shiitake mushrooms and snow peas in garlic-infused oil in another pan.
He held up to the camera a white plate that held a square piece of meat. It had less red tint and more brown overtones than a typical slab of raw flesh, but it looked steaklike nonetheless. He sprinkled it with salt and pepper and then poured some oil into the pan and waited for it to start smoking.
6% of the U.S. population now identifies as vegetarian, an increase from 1 percent just in the past few years.
At the start of the demonstration, I asked Ilan if there was anything about this meat—from the type of cells they used to grow it to how it was formed in the lab—that would make it difficult to cook. Quite the opposite, he said: “We don’t have the problem of a cow [being raised] outdoors, which can toughen the meat. We create the perfect steak in the lab from the start, according to the flavor, texture, and structure we want.”
He dropped the square onto the sizzling cast iron, then swirled the vegetables some more. Once he was happy with the steak’s color, he flipped it; then he dribbled a wine reduction onto a plate that was already decorated with sprigs of rosemary. Using tweezers, he placed the steak in the center of the dish and surrounded it with a few snow peas and shiitakes. He held up the finished arrangement, which looked very much like something that would be served at a good restaurant.
Purported benefits notwithstanding, Aleph and its competitors are going to need to overcome a lot of resistance to get Americans to replace their filets mignons and pork chops with ones grown in a bioreactor. First, the price will need to fall dramatically. Aleph says it can produce a steak for $50, but considering that Memphis Meats’ hamburger was $6,000 per pound just four years ago and steak is a lot more complicated, that quote seems suspiciously low. The FDA and the Department of Agriculture will need to approve the products in a complicated two-step process that has never been tried.
 An old-school cheeseburger. DA-KUKGETTY IMAGES
An old-school cheeseburger. DA-KUKGETTY IMAGES
And then there’s the question of how fake steak will go over with the people who patronize the world’s top restaurants. “My initial impression is that this is undoubtedly good for the planet and bad for gourmands and foodies,” says the novelist Jay McInerney, who writes about food and wine (the latter for Town & Country). “It’s a noble effort, but it’s hard to imagine that, certainly in the early stages, it will be relevant to people who take food really seriously. It seems like a mass-market product, and a tough sell for people who dine at Per Se.”
The startups will also need to allay the suspicions of what has been called the “food movement”—those who favor the artisanal and eschew the industrial. Plant-based imitation burgers such as those from Beyond Meat and Impossible Foods have faced opposition for being overly processed. Not coming from any heretofore known method of producing food, cultivated meat is likely to bear similar scrutiny.
 CRISTINA PEDRAZZINI/SCIENCE PHOTO LIBRARYGETTY IMAGES
CRISTINA PEDRAZZINI/SCIENCE PHOTO LIBRARYGETTY IMAGES
“We don’t need this,” says chef and author Mark Bittman, whose most recent book, Animal, Vegetable, Junk: A History of Food, from Sustainable to Suicidal, details some of the less savory trends in recent culinary habits. “We have perfectly good food out there” to replace environmentally damaging, unhealthful beef, “and it’s called plants.” To convince food movement advocates, he says, cultivated meat would need to fulfill its promises of safety and low resource use and reduce consumption of animal products, rather than merely add another option to the menu.
More than anything, though, the new companies will need to produce something that is as good as the meat you can buy in a store, or a steak you’d order at a restaurant. Suzanne Tracht, chef and owner of Los Angeles’s Jar—consistently rated among the best steakhouses in the U.S.—told me, “I have a chophouse, so I have to source and get the best. My customers are not asking, ‘Where did this piece of meat come from?’ But they do know what tastes good.”
Where’s the Beef?
Aleph’s steak grew out of a process developed at the Technion-Israel Institute of Technology by Shulamit Levenberg, an expert in biomedical engineering who hopes to cultivate from cells human tissue for transplant. She worked for years at MIT with Robert Langer, a pioneer in the field of tissue engineering, before co-founding Aleph with Didier Toubia, a life science entrepreneur.
Growing muscle tissue for human consumption is much simpler than growing it for transplant. Transplanted tissue requires functional cells, such as muscle cells that can contract. But for meat, Lavon says, “We just need the components that bring the unique taste.” A “bioprinting” technique that Levenberg helped develop enables the steak to grow three-dimensionally, with a structure similar to what is cut from a slaughtered cow, and in a single batch, with muscle, fat, and other types of tissue maturing together so they’re integrated from the outset, just as nature does it. Other companies grow different cells separately and combine them later, so all they can come up with is less refined meat, such as hamburger or chicken nuggets.
Aleph starts with a small sample from an animal (a Holstein cow, for the ribeye prototype) acquired in a nonsurgical procedure that provides enough cells for thousands of tons of meat. These cells can grow into any type of cell: muscle, fat, skin, whatever. After all the types have been replicated in an off-the-shelf bioreactor, they are 3D-printed into the combination Aleph wants. “We are designing our meat,” Lavon says, “so we can choose the proportion of fat cells to muscle cells.” The combination is then grown in a medium Aleph developed that mimics blood serum in its composition of vitamins, amino acids, minerals, fatty acids, proteins, and sugars.
10% of humanity’s greenhouse gas emissions come from beef and milk production (more than 2/3 the amount produced by the transport sector).
Less fat means less cholesterol. More of a particular compound in the growth medium means more omega-3 polyunsaturated fatty acids, which have been shown to reduce heart attacks, particularly among people who don’t eat much fish. Luining, of Meatable, foresees a day when “you can have meats that are beneficial for certain groups—I can even imagine personalized nutrition.”
Beyond Meat and Impossible Foods, which have wowed critics and consumers with their plant-based burgers’ remarkable resemblance to the real stuff, spent years figuring out how to win over carnivores by replicating the taste, texture, and aroma of meat. One component of beef that is important to the experience of both eating and cooking it, they found, is myoglobin, a type of protein found in muscle cells. Each company needed to come up with a means of mimicking myoglobin’s function in the sensory experience of biting into a cut of beef. But Aleph, Meatable, Memphis, and the rest don’t need to replicate the components of meat; myoglobin—and the 1,000-plus other molecules that comprise our interaction with food from animals—is already in the product. Because the product is, biologically, meat.
Big Meat’s Big Beef
“Traditional” beef enjoys several built-in advantages that will make it difficult for lab-grown meat to replace it in shopping carts in wealthy countries, and without the early support of Americans and Europeans, it will be difficult for the fledgling industry to grow to the scale necessary for it to furnish the expected demand for meat from the developing world. First, in the U.S. the federal government leases land to ranchers to graze cattle at below-market prices. The subsidies make meat cheaper, inducing Americans to eat more of it than anyone else on earth.
Then there’s the beef lobby, which doesn’t even want cultivated beef to be called beef. Missouri and several other states have already passed legislation banning companies like Aleph from using the word. Since the United States Cattlemen’s Association’s members pay for the organization to promote “beef,” “it would seem unfair to beef producers to have this new player in the game come in to take advantage of the good nutritional reputation beef has built up,” says Maggie Nutter, a Montana rancher who chairs the association’s labeling committee.
 PHOTOGRAPH TAKEN BY ALAN HOPPSGETTY IMAGES
PHOTOGRAPH TAKEN BY ALAN HOPPSGETTY IMAGES
As Bittman suggests, it may come down to the environmental claims, which have been at the center of the new industry’s portrayal of itself to investors and media. “There really needs to be independent analyses normalized among different products, so consumers can have confidence that if someone wants to buy something more sustainable, it really is, on some objective level,” says Gregory Jaffe, director of the Project on Biotechnology at the Center for Science in the Public Interest, a DC-based nonprofit.
Regardless, the potential health benefits may be enough to convince some hard-core meat eaters. Suzanne Tracht notes that her restaurant in Los Angeles is “down the street from the heart clinic at Cedars-Sinai, so I think there will be people interested” in cholesterol-free beef. While she’s skeptical that cultivated meat will be as savory as the cuts she procures from small operations, she’s curious.
“I’m looking forward to trying it with the real thing, side by side,” she says.


 Aleph Farms co-founder Didier Toubia holds a plate with a “traditionally” grown steak in one hand and petri dish containing cow cells in the other. PICTURE ALLIANCE
Aleph Farms co-founder Didier Toubia holds a plate with a “traditionally” grown steak in one hand and petri dish containing cow cells in the other. PICTURE ALLIANCE ILLUSTRATION BY JOE DARROW
ILLUSTRATION BY JOE DARROW An old-school cheeseburger. DA-KUKGETTY IMAGES
An old-school cheeseburger. DA-KUKGETTY IMAGES CRISTINA PEDRAZZINI/SCIENCE PHOTO LIBRARYGETTY IMAGES
CRISTINA PEDRAZZINI/SCIENCE PHOTO LIBRARYGETTY IMAGES PHOTOGRAPH TAKEN BY ALAN HOPPSGETTY IMAGES
PHOTOGRAPH TAKEN BY ALAN HOPPSGETTY IMAGES
 Prof. Moshe Shoham with the Mazor Robotics system, which was sold to Medtronic for $1.64 billion (Courtesy)
Prof. Moshe Shoham with the Mazor Robotics system, which was sold to Medtronic for $1.64 billion (Courtesy)


 Eytan Stibbe is the second Israeli astronaut to launch into space. Seen here at a press conference in Tel Aviv on May 5, 2021. (Flash90)
Eytan Stibbe is the second Israeli astronaut to launch into space. Seen here at a press conference in Tel Aviv on May 5, 2021. (Flash90)
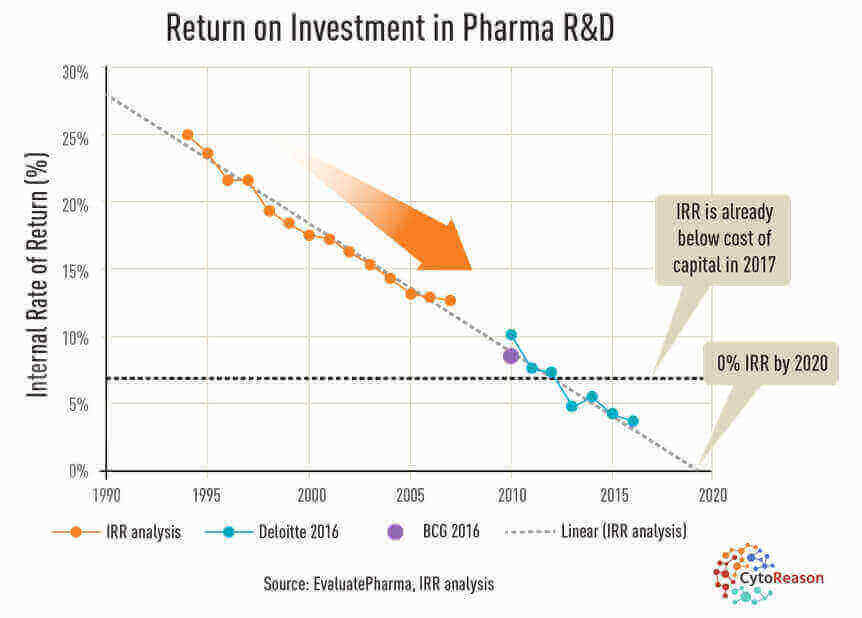 Returns on developing new drugs are being outpaced by rising costs. (EvaluatePharma via CytoReason)
Returns on developing new drugs are being outpaced by rising costs. (EvaluatePharma via CytoReason)