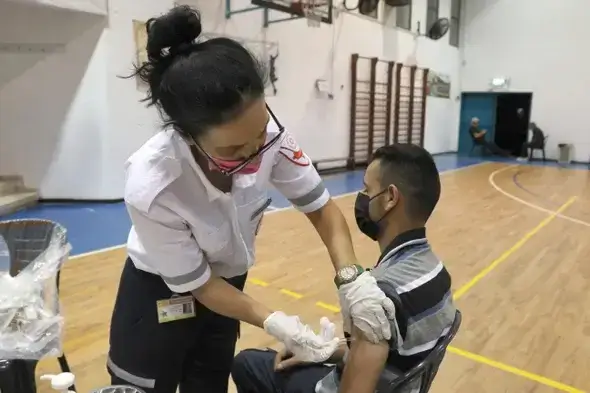
The risk of severe disease dropped by factor of almost 20 in people over 60—but some dispute the benefits of offering an additional dose
Older Israelis who have received a third dose of a COVID-19 vaccine are much less likely to test positive for SARS-CoV-2 or to develop severe COVID-19 than are those who have had only two jabs, according to a highly anticipated study published on 15 September.
The standard regimen for messenger RNA-based COVID-19 vaccines is two doses, but some governments, including Israel’s, have started administering third ‘booster’ shots. The latest study evaluated 1.1 million Israelis over the age of 60 who had received their first two doses at least five months earlier. Twelve or more days after receiving a third jab, participants were about 19.5 times less likely to have severe COVID-19 than were people in the same age group who had received only two jabs and were studied during a similar time period.
“It’s a very strong result,” says Susan Ellenberg, a biostatistician at the University of Pennsylvania in Philadelphia, who adds that the data might be the most robust she has seen in favour of boosters. But potential biases in the data leave some scientists unconvinced that boosters are necessary for all populations—and the data do not dispel concerns about vaccine equity when billions of people are still waiting for their first jab.
Israel, which got an early start on vaccinating its population, began offering third doses of the Pfizer–BioNTech vaccine in July, to people aged 60 and over. The latest analysis links the third jab not only with a significant reduction in severe COVID-19, but also with an 11.3-fold reduction in SARS-CoV-2 infections.
But Ellie Murray, an epidemiologist at Boston University in Massachusetts, cautions that observational studies such as this analysis can contain biases that are difficult to identify and account for. For example, people who sign up to get a booster might have a different risk of COVID-19, or behave differently, from people who do not get a third jab.
Ellenberg says that the authors try to address some of these potential biases. Even if not all biases have been eliminated, she says, the magnitude of the effect suggests that the booster offers some protection, at least in the short term. The authors of the study could not be reached before publication.
GLOBAL RAMIFICATIONS
The findings come as a slew of wealthier nations consider offering booster shots. An advisory committee of the US Food and Drug Administration (FDA) will discuss Pfizer’s application to supply boosters in the United States on 17 September. One of the authors of the Israeli study is slated to present data to the committee.
Murray argues that the potential biases in the data, and insufficient evidence for waning immunity after vaccination, mean that the latest findings don’t indicate a “strong need” for boosters. “From a public-health perspective, it’s way, way more impactful to get more people vaccinated than it is to boost the vaccine effectiveness by a few percentage points in those who have already gotten the vaccine,” she says.
Murray is not alone in finding the Israeli results insufficient to justify boosters. A review published on 13 September by a team that includes two high-ranking FDA scientists cites a preprint of the study and notes that the short-term protective effect documented in Israel “would not necessarily imply worthwhile long-term benefit”.
Dvir Aran, a biomedical data scientist at Technion—Israel Institute of Technology in Haifa, says that Israel has deployed boosters to stop transmission in younger people and to prevent severe disease and deaths in older people.
“Is it the best way? Whether a two-week lockdown would have given a similar result, I can’t answer that question,” he says. “But it’s an interesting approach, trying to stop an outbreak like this with vaccinations.”
Study: COVID booster recipients 20 times more protected against serious illness
As US officials set to mull okaying Pfizer’s 3rd dose, data from a million Israelis shows it boosts protection from infection tenfold compared with eligible people who got 2 shots
A new study conducted in Israel shows that individuals given a third COVID-19 vaccine dose are nearly twenty times more protected against serious illness and more than ten times more protected against infection, compared with those who received their second dose at least five months previously.
The research, published on Wednesday by The New England Journal of Medicine, showed that 12 days after receiving a booster shot of a Pfizer-BioNTech COVID-19 vaccine, the chance of infection was 11.3 times less than among those eligible for a third shot but didn’t get one.
And the chance of suffering serious illness as a result of COVID-19 among those who had received a booster shot was 19.5 times less, the research said.
The study was conducted by researchers from the Weizmann Institute of Science, the Ministry of Health, the Technion, the Hebrew University, Sheba Medical Center, and the KI Institute.
Even with a more conservative analysis, which attempted to control possible behavioral differences between the two groups, the infection rate was at least 5 times lower in the group that had received the booster shot, the Health Ministry said in a statement.
The research includes data from more than 1 million Israelis. Among those who hadn’t received a booster shot despite being eligible, there were 4,439 confirmed infections, including 294 serious patients. Among those who received the booster at least 12 days previously, there were 934 infections including 29 serious cases.
The Israeli data could not say how long the boosted protection lasts.
But a separate study conducted at Sheba Medical Center in Ramat Gan, outside Tel Aviv, has stoked optimism as to the amount of time for which the booster shot retains its protection.
The study found that the antibody levels a week after the third COVID-19 vaccine dose was administered to its staff were ten times higher than their levels a week after the second dose was administered.
Israel — the first country to officially offer a third dose — began its COVID-19 booster campaign on August 1, initially rolling it out to those over the age of 60. It then gradually dropped the eligibility age, eventually expanding it to everyone aged 12 and up who received the second shot at least five months ago.
As of Thursday, nearly 3 million Israelis had received their third dose.
Meanwhile in the US, influential government advisers will debate Friday if there’s enough proof that a booster dose of Pfizer’s COVID-19 vaccine is safe and effective — the first step toward deciding which Americans need one and when.
The Food and Drug Administration on Wednesday posted much of the evidence its advisory panel will consider.
Pfizer’s argument is that while protection against severe disease is holding strong in the US, immunity against milder infection wanes somewhere around six to eight months after the second dose.
More important, Pfizer said, those antibodies appear strong enough to handle the extra-contagious Delta variant that is surging around the world.

To bolster its case, Pfizer pointed the FDA to the new data from Israel.
Pfizer said the data published on Thursday translates to “roughly 95% effectiveness” against Delta — comparable to the protection seen shortly after the vaccine’s rollout earlier in the year.
In Israel, the R-value — the reproduction rate of the virus measuring the average number of people each positive person infects — rose to 1.14 on Thursday, after it had hit a 4-month low of 0.81 just days earlier.
Any number over 1 indicates infections are rising, while a figure below that signals that an outbreak is abating.
There were 8,601 new COVID-19 cases diagnosed on Wednesday, according to the Health Ministry.
Of the 83,704 active cases, 654 are in serious condition. Since the start of the pandemic last year, 7,465 people have died of COVID-19 complications in Israel.
